Get traceability, tracking, and flow control
Track & Trace maximizes the quality of your assembly line and gives you full traceability on your products and sales orders.
With a Track & Trace solution from CIM, all components are marked with unique serial numbers/bar codes, and customized flow control ensures that components go through all defined process steps in the right order. The detail level of traceability and flow control is entirely defined by your needs.
Pharma customers look for serialization with full compliance. We guarantee compliance to all international standards and our solutions are ideal for retrofit as well as new systems.
The Benefits include:
- Full traceability in your products and sales orders
- Visibility of service and repair needs in case of faulty parts
- Improved quality control through the use of flow control
- Documented quality through consolidated test reports
- Full integration with your existing business management system (ERP system)
The future of Track & Trace lies in the ability to identify each individual object in a production. We are highly experienced in these advanced Track & Trace solutions because of our strong track record in pharma packaging where it is mandatory to individually track the smallest sellable unit.
See more about our products down below or visit our Pharma Website:
Serialization solutions for pharma
Compliance is everything in serialization. CIM Pharma delivers solutions that comply with all international standards. Our serialization solutions are built on tried-and-tested software to make sure we can guarantee our customers:
- Integration with any existing software or hardware
- Minimal effect on OEE
- Compliance with all international standards, including CFR21 part 11
- Solutions ideal for retrofit as well as new systems
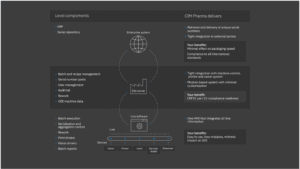
Covering all levels of packaging
We cover all levels of the packaging hierarchy as well as all levels of dataflows. That means our customers only have to shop in one place to get a solution that provides everything they need.
- Works at device, line, and site level
- All levels of the packaging hierarchy covered: from product to carton to shipper to pallet
- Full integration and connection with relevant dataflows from level 1-4
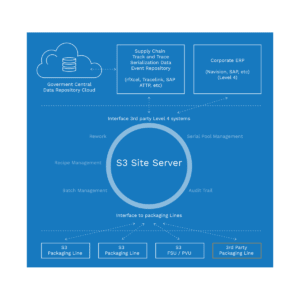
Track & Trace made simple
The CIM S3 Line Software is a stand-alone software solution that makes Track & Trace as simple as possible. It can be connected to the CIM S3 Site Server, other third-party level 3 servers, and to Level 4/5 Cloud based systems. S3 handles the batch, serial number and all other data communication with printers, lasers, vision systems etc. on the line.
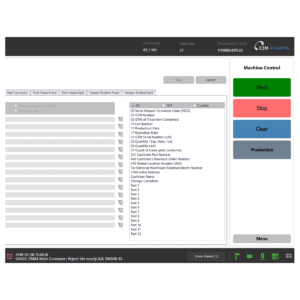
Features of CIM S3
- Hardware support of a wide range of printers, vision systems, PLC systems, etc.
- Batch Execution and reporting
- Smart user Management
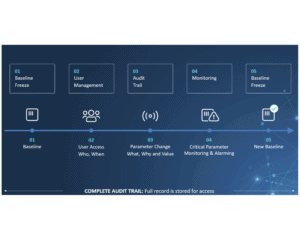
- Audit Trail on user action, recipe changes, rework etc.
- Serial Pool Management
- Rework: Decommissioning of serialized items
Get everything printed on the production line
Integrate printing into your production line for reduced batch change-over-time, maximum flexibility, and improved quality control. With digital printing, data is fed into the printer from a label artwork database and a batch information database. That means the complete label content – both the static artwork, text, and dynamic data – is printed at once and each label can be inspected individually.
This results in a number of benefits:
- No or little batch change over time
- Minimal Line Clearance
- Minimal time for Line Validation
- Improved OEE
- Maximum flexibility
- Full label inspection of every label
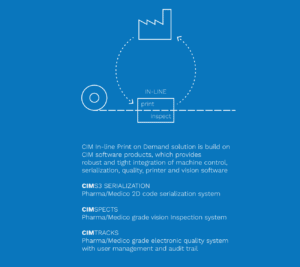
A plug-and-play solution for in-line printing
CIM offers a fully tested plug-and-play solution for digital in-line printing that ensures complete compliance with all regulations. Our software is built on standard validated technology and includes well-known CIM products you already know.
Keep Track and Stay Compliant
For production companies, keeping track of the activities on assembly lines and packaging lines is key to staying compliant with international standards and regulations. This is especially true for the pharma industry, where the CFR 21-part 11 regulation sets high standards for electronic documentation and signatures. Failure to comply means failure to operate, and that is devastating to business. So choosing the right audit trail system is more important than ever.
CIM TRACKS acts as a smart bridge between machine components at level 1 and the Enterprise System at level3 and allows you to monitor critical parameters and perform user management much more efficiently.
References
Don't take our word for it
Henrik Just
Project Manager Serialization
Stevanato Group
Jan Bigum
Lead Engineer
Grundfos Production IT
Andy Cumming
Consultant in Manufacture Change


Contact our specialist in Track & Trace Solutions
Anders Meister, Chief Sales Officer