Vision inspection for smart factories
The SPECTS vision inspection system is designed for inline inspection on high-speed assembly and packaging lines and comes as an all-in-one solution with software and cameras.

There is more to a vision system than inspection
Vision inspection is a key process for quality assurance in pharmaceutical manufacturing. However, most traditional systems have a hard time keeping up with the industry’s demands for process insights and efficiency. Common challenges include:
- Scattered GxP data.
- Manual document handling.
- No tracking of critical parameters.
- Difficult to adapt to new demands and technologies.
- Slow and resource-demanding commissioning and validation.
SPECTS is made for pharma
The SPECTS vision inspection system helps pharmaceutical manufacturers automate document and GxP data handling.
As a modular, pre-validated GAMP 5 Cat. 4 vision inspection system, SPECTS offers CFR 21 part 11 compliance and easy implementation.
Efficient vision inspection management
Efficient inspection management requires more than just camera and lighting designs, especially when GxP data is involved.
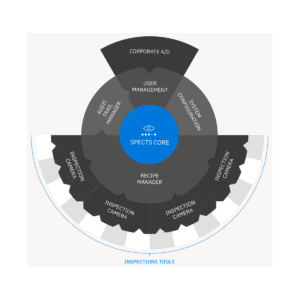
SPECTS is a comprehensive black box solution that automates document and GxP data handling and integrates seamlessly with level 2 and 3 systems.
The SPECTS solution encompasses all aspects of inspection management, from handling recipes to tracking critical parameters and generating batch reports.
Built for streamlined implementation
As a pre-validated black box, SPECTS allows pharma companies to test and validate only inspections and quality-related parameters in connection with a FAT, not the entire system.
SPECTS is GAMP cat. 4 configurable software, whose core functionalities exist alongside customizable behaviors. Configuration is done through intuitive user interfaces.
Because the pre-validated core remains untouched, pharma companies can trust the core functionality of the system to operate reliably and focus only on testing configurations.
This makes installation qualification, operational qualification, and process qualification much more efficient. The simplified process also makes it easier for pharma companies to adopt new technologies.
Explore features and functionality here
SPECTS replaces logbooks, paper forms and manually generated PDF’s by automating document handling and GXP data flow.
Our user management integrates seamlessly with Active Directory, automatically generating audit trails on all machines. This eliminates concerns about handling audit trails or logins and makes it possible to recreate the history of the machine in minutes.
SPECTS automatically exports critical audit trails to any level 2/3 system. This ensures that events occurring during or between batches are documented in an overarching system and allows for more continuous monitoring.
SPECTS is designed as a modular platform with a pre-validated core surrounded by standard tools and plugins. This means that it is open for extensions and easily adaptable while keeping core functionality intact.
The modular architecture means that unique requirements not supported by the platform’s standard tools, such as data delivery to a specific level 3 system, can be met through the creation of separate modules and interfaces that can be implemented as plugins and validated independently.
The standard inspections in SPECTS cover all common use cases. Custom inspections can easily be added to the platform.
Standard inspections include:
- Device inspection
- Mix-up test
- 360 degree inspection
- Label inspection
- Label placement
- Glass inspection
SPECTS comes with a full documentation package.
We also assist with writing and reviewing test plans.
Analyze inspection results and corresponding images, investigate outliers and spot trends in SPC data.
Our powerful production data platform CATCH.AI provides you with the insights you need to improve processes and take preventive action.
As SPECTS can integrate with any type of PLC using Beckhoff hardware and standard industrial protocols, it is easy to control the vision system from the PLC.
SPECTS can execute pre-trained Deep Learning models through e.g. Halcon, Pytorch, YOLO and ONNX.
As a standard we provide Beckhoff controllers, but as SPECTS allows for complete flexibility in the choice of industry-grade controllers, cameras and lighting, we can create a solution that meets specific customer demands and project requirements.
As all image processing happens in SPECTS, cameras can be of any industry-grade brand and easily replaced.
The SPECTS vision inspection system gives you a number of benefits
- Improves efficiency by automating tasks and reducing manual processes.
- Securely manages GxP data and documents critical activities.
- Increases utilization of machinery by reducing BCO time.
- Supports all regulatory requirements in a documented manner.
- Makes implementation a breeze with less time spent on test and validation.
- Adapts easily to specific needs and new technologies – now and in the future.
- Runs reliably in operation – no nasty surprises.


Straight from the source
What our customers say about the SPECTS vision inspection system
Henrik Just
Project Manager Serialization
Stevanato Group
Jan Bigum
Lead Engineer
Grundfos Production IT
Jan Rheinholdt Mohrdieck
Technical Chief
SBS Friction A/S
CASES
Transformational stories from our customers
Want to manage inspections efficiently?